Latest Research
- 2026.05.01
- Fukushima-Shoji Group
Synthesis and Dual Fluorescence Behavior of Higher Heteroacenes with Periodically Incorporated Boron Atoms
Incorporation of boron atoms into π-conjugated systems enhances their electron-accepting character and markedly modifies their electronic structures and photophysical properties, owing to the low electronegativity of boron and the presence of vacant p orbitals. These features make boron-containing π-systems highly attractive for the design of organic electronic and photofunctional materials. Based on our concept of "vacant orbital engineering," we have developed new organoboron compounds by tailoring the bonding environment and spatial arrangement around boron centers, thereby bringing out their intrinsic potential.[1]
The effects of boron incorporation described above are expected to become more pronounced as a greater number of boron atoms are introduced into a single π-conjugated framework. However, such boron-rich systems remain scarce, largely due to challenges associated with their synthesis and chemical stability.
In this study, we focused on diboraanthracene, a molecule in which two carbon atoms in the anthracene framework are replaced by boron atoms (Fig. 1). By designing a synthetic strategy to construct this motif, we envisioned that π-conjugation could be extended in a one-dimensional manner through boron-bridged structures, leading to a new class of heteroacenes.[2] After careful optimization of the reaction conditions, we successfully synthesized a heptacene derivative incorporating six boron atoms (B6-hept, Fig. 1). To the best of our knowledge, this compound represents the most boron-rich heteroacene reported to date. In addition, the reaction also afforded shorter homologues, including a tetraborapentacene derivative (B4-pent) and diboraanthracene (B2-ant).
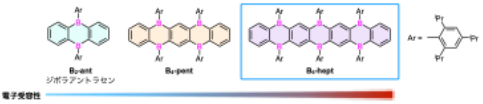 |
||
| Fig. 1. | Boron-containing heteroacene derivatives developed in this work. | |
The electronic properties of this series of compounds were investigated, revealing, as anticipated, that the electron-accepting ability increases progressively with the number of boron atoms incorporated. We then examined their photophysical behavior and found that only B6-hept exhibits pronounced dual fluorescence. Detailed analyses combining spectroscopic measurements under various conditions with theoretical calculations indicated that this behavior originates from dynamic conformational interconversion of B6-hept in solution. Such conformational dynamics are attributed to steric interactions between the bulky substituents introduced at the boron centers to ensure chemical stability. In contrast, dual fluorescence is only marginally observed for the shorter π-system B4-pent and is completely absent in B2-ant. These results demonstrate that the combination of π-system extension and steric effects of the substituents induces conformational dynamics, which in turn gives rise to the unique emission behavior.
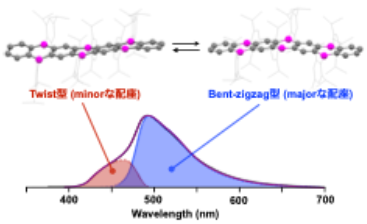 |
||
| Fig. 2. | The conformational isomers and dual fluorescence behavior (λex = 355 nm) of B6- | |
The findings of this study demonstrate that steric and conformational design provides an effective strategy for controlling emission properties in boron-containing π-conjugated systems, and thus opens new possibilities for the development of responsive functional materials.
| 参考文献 | |
| [1] | Shoji, Y.; Kashida, J.; Fukushima, T. Chem. Commun. 2022, 58, 4420 (DOI: 10.1039/D2CC00653G). |
| [2] | Yokochi, T.; Yokoyama, H.; Tsukada, T.; Sakai, H.; Hasobe, T.; Fukushima, T.; Shoji, Y. Angew. Chem. Int. Ed. 2026, e3365679 (DOI:10.1002/anie.3365679). |



